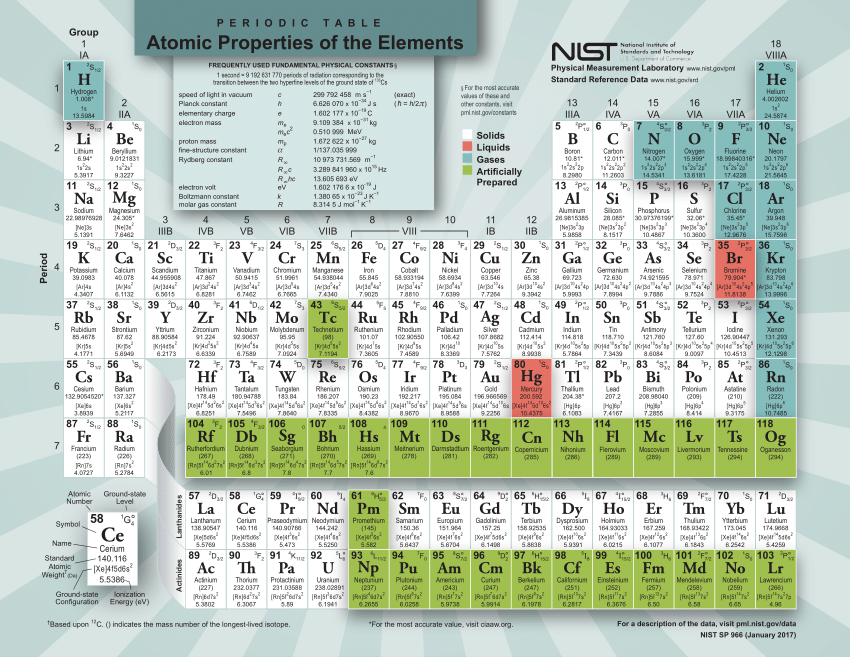
A row on the periodic table is called a period. Heres a nice Periodic Table and more pdf for you to use for this class. For instance, the relative atomic mass of zirconium (Zr) is 91.22. This is usually located under the symbol and name of the element. To find an element’s molar mass, start by checking the periodic table for the relative atomic mass of the element. 
Now we interpret the coefficients as referring to molar amounts, not individual molecules. The periodic table is your ultimate conversion chart for converting any substance into another substance and doing so with exact proper amounts (masses and moles). Molar mass is the mass in grams of 1 mole of any given substance. Please note that the elements do not show. The unity for atomic mass is gram per mol. Interactive periodic table with up-to-date element property data collected from authoritative sources. 2 For normal samples from earth with typical isotope composition, the atomic weight can be approximated by the standard atomic weight 3 or the conventional atomic weight. The lightest chemical element is Hydrogen and the heaviest is Hassium. By adding the atomic masses taken from the periodic table, the molar mass can be determined. The periodic table provides the mass of each individual element, denoted beneath the element's symbol. The molar mass of each element is the same as the atomic mass listed on the periodic table. Molar mass is calculated by adding the atomic masses of a given compound. We can leave out the word mol and not write the 1 coefficient (as is our habit), so the final form of the equation, still balanced, is The molar mass of atoms of an element is given by the relative atomic mass of the element multiplied by the molar mass constant, Mu 1 × 103 kgmol1. The molar mass is the mass in grams of one mole of a substance. But 6.022 × 10 23 is 1 mol, while 12.044 × 10 23 is 2 mol (and the number is written that way to make this more obvious), so we can simplify this version of the equation by writing it as These coefficients also have the ratio 2:1:2 (check it and see), so this equation is balanced.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
