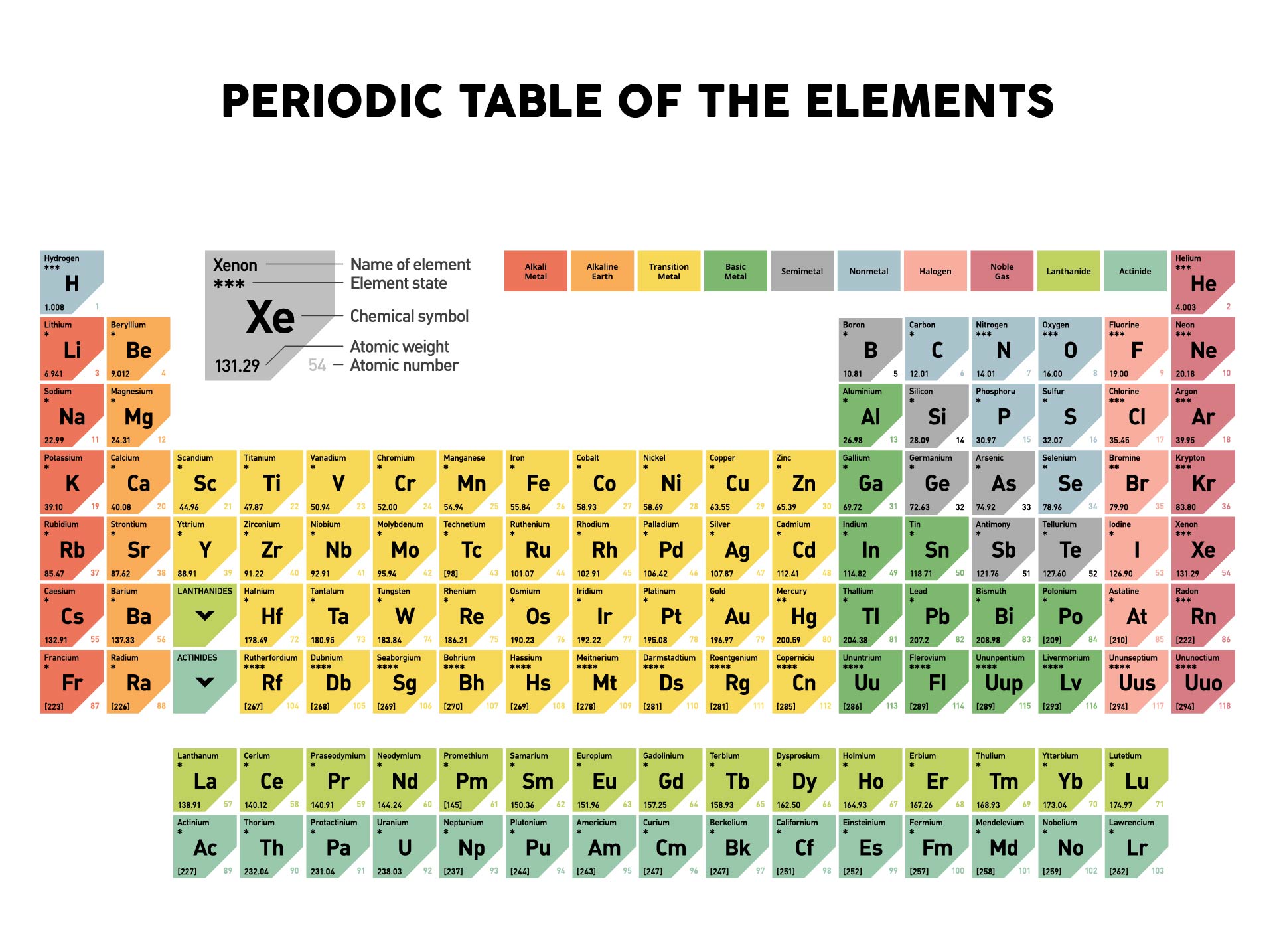
Please re-write this article to improve it to explain concepts before they are used in answers or taken for granted as prior knowledge when they have not first been defined in Unit 1. Interactive periodic table with all properties. Formerly known as Standard Reference Database (SRD) 145, but reclassified as an. Access the Table: PDF without crop marks PDF with crop marks. The PDF is suitable for high-resolution color printing for desk or wall-chart display. Know all element Facts, Electronic Configuration, oxidation state, isotopes. The periodic table contains NIST’s latest critically evaluated data for atomic properties of the elements. Upto Date latest full Modern Periodic Table with dynamic layouts. The problems are then for ionic compounds that are not monoatomic. Dynamic Interactive Periodic Table of Elements with names, symbols, atomic mass - Chemical, Physical, Atomic and Thermal Properties & Trends. Transition metals form ions with different charges. acetate PERIODIC TABLE OF IONS arsenate arsenite benzoate borate bromate carbonate chlorate chlorite chromate cyanate cyanide dichromate CH3COO AsO4 3 AsO3 3 C6H5COO BO3 3 BrO3 CO3 2 ClO3 ClO2 CrO4 2 CNO CN Cr2O7 2 oxalate perchlorate periodate permanganate peroxide phosphate pyrophosphate sulfate. The use of magnitude is used without being introduced followed by a statement that isn't well described, 'The magnitude of the charge is equal to the number of electrons lost, which is equal to the number of valence electrons in the neutral atom.'Īluminum is in group 13, or IIIA is stated when no introduction has been provided to what IIIA means. The periodic table helps to categorise the known elements and make predictions about ones that we haven’t yet discovered. 
Where in Unit 1 is s, p, d, f and shell and subshell defined prior to this article. IUPAC group number is also not described, it is simply used, without definition or prior description.Įlectron configuration is used as part of a hidden explanation as an answer, yet the article or Unit 1 does not explain what an electron configuration is. Where in Unit 1 is Octet and Valence given definition. Octet is used without definition, followed by valence also being used without definition. 
We are being asked to 'Remember that periodic groups refer to columns on the periodic table, whereas rows are known as periods' when this concept has also not been initially defined in this article or in Unit 1. Where is monatomic defined in this article or in Unit 1. Words are used without first having had a definition or explanation in the article or earlier in Unit 1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
